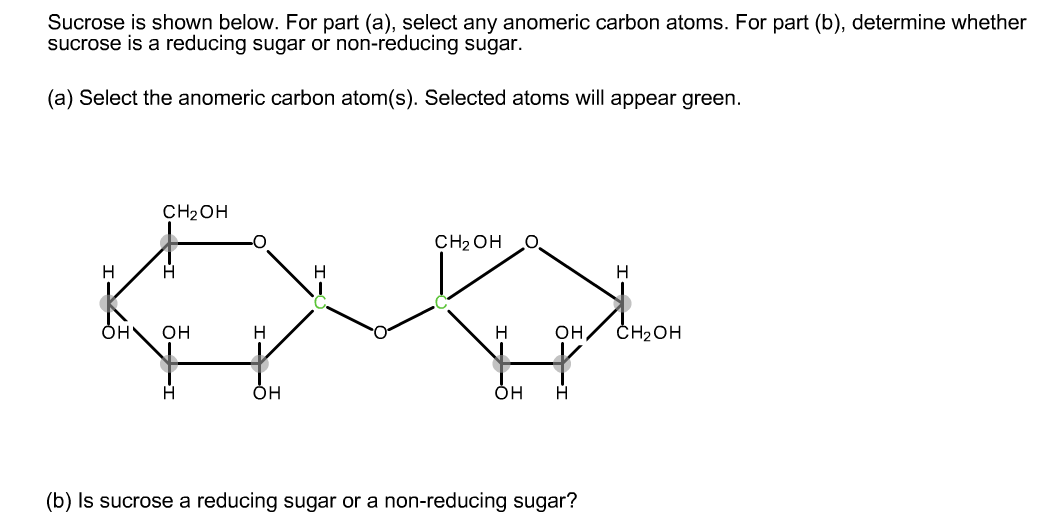
This sets us up to draw the C 5-OH attacking the carbonyl carbon, forming a new O–C 1 single bond and breaking the C 1-O pi bond.Īfter proton transfer, we now have one of two rings, which we can depict as Haworth projections! I also drew the C 2-C 1 bond dashed as well. In the figure below I drew it with a “dashed” bond, owing to the tetrahedral geometry on the carbon. So we’ll make the following three “moves”:Īfter rotation, the C 5-OH is on the side rather than pointing down. Interchanging any three groups on a carbon does a bond rotation. It’s helpful to perform a bond rotation on the C 5 carbon to make the stereochemistry on the ring clearer. This sets us up to form a bond between the C 5-OH and the carbonyl carbon (C-1), which will make a new ring. Next, let’s turn this molecule on its side, 90 degrees clockwise. The first step in converting a Fischer to a Haworth is to draw in these wedges and to number the carbons. One way this was taught to me was to remember that “ the arms come out to hug you“. īy convention, the horizontal bonds on a Fischer projection actually point out of the page. We’ll start by remembering what the Fischer projection really represents.Īlthough all the bonds in the Fischer might be drawn “flat”, it’s not meant to be understood that way!. hexagonal) depiction in the Haworth projection – the “long way”. Let’s start with the example of converting D-glucose drawn in a Fischer projection to a pyranose (i.e. (We’ll show how to deal with furanoses too.) Well, there are a couple of tricks, and that’s what this post is about. 
(the reverse question can be asked too: “convert a sugar drawn in a Haworth to an open-chain Fischer. “Convert this (sugar) from the Fischer projection to a cyclic pyranose form as a Haworth projection.” Here’s a relatively common problem in the realm of sugar chemistry: Converting A Fischer to a Haworth (The Long Way)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
